Emgality (gacanezumab-gnlm) solution has been FDA approved for treatment of episodic cluster headache in adults, the first of its kind.
Emgality was approved in 2018 to prevent migraines in adults, and now has shown to reduce the frequency of cluster headache attaches during a clinical trial of 106 patients.
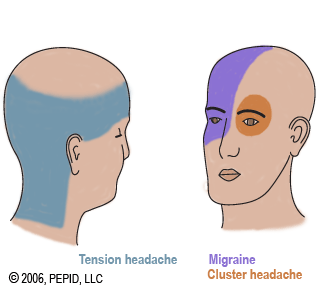
“Emgality provides patients with the first FDA-approved drug that reduces the frequency of attacks of episodic cluster headache, an extremely painful and often debilitating condition,” said Eric Bastings, M.D., deputy director of the Division of Neurology Products in the FDA’s Center for Drug Evaluation and Research. “The FDA is committed to continuing to work with drug developers to bring treatments for unmet medical needs to patients.”
Read the full FDA Press Release here.
[elfsight_social_share_buttons id=”3″]

